How many rechargeable lithium-ion batteries are you wearing? How many are in your general vicinity?

Probably more than a few, and they’re great for powering all the things important to modern lives: cellphones, watches, computers, cars and so much more.
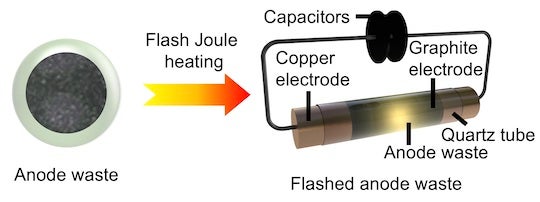
But where they go when they fail is a growing problem. Rice University scientists believe they have a partial solution that relies on the unique “flash” Joule heating process they developed to produce graphene from waste.
The Rice lab of chemist James Tour has reconfigured the process to quickly regenerate graphite anode materials found in lithium-ion batteries, removing impurities so they can be used again and again.
The lab’s work appears in Advanced Materials.
Flashing powdered anodes from commercial batteries recycles some of what the researchers called the “staggering” accumulation of waste they currently leave behind. In just a few seconds, a jolt of high energy decomposes inorganic salts including lithium, cobalt, nickel and manganese from an anode. These can be recovered by processing them with dilute hydrochloric acid.
“The production of lithium-ion batteries in 2026 is expected to be five times what it was in 2017, and right now, less than 5% of them are recycled,” said Tour, who introduced the flash process for graphene in 2020. “That puts a heavy load on the environment, as these spent batteries are processed and the anodes burned for energy or sent for landfills.
“We’re claiming our process can recover critical metals and recondition anodes in a far more environmentally and economically friendly manner,” he continued.
The lab reported that flashing anodes degrades the solid-electrolyte interphase (SEI), which conducts lithium ions but also insulates the anode from detrimental reactions.
Flashing then coats the remaining graphite particles with an ion-permeable carbon shell that contributes to their future capacity, rate performance and cycling stability compared to materials conventionally recycled in a time-consuming and energy-intensive process known as high-temperature calcination.
The lab estimated it would cost about $118 to recycle one ton of untreated anode waste. They demonstrated that flash-recycled anodes have a recovered specific capacity of 351 milliAmp hours per gram at 32 degrees Fahrenheit, superior to the rate performance and electrochemical stability of untreated or calcinated recycled anodes.
The recycled, flashed anodes the researchers tested retained more than 77% of their capacity after 400 recharge cycles.


“Beyond the spent graphite anodes, we are confident that the cathodes, the electrolytes and their mixtures can be effectively recycled or reconditioned by our method,” said Rice graduate student Weiyin Chen, lead author of the study.
Co-authors are Rice academic visitor Rodrigo Salvatierra; alumnus John Tianci Li; research scientist Carter Kittrell; graduate students Jacob Beckham, Kevin Wyss, Nghi La, Paul Savas, Chang Ge, Paul Advincula, Phelecia Scotland and Lucas Eddy; and postdoctoral researchers Bing Deng and Zhe Yuan.
Tour is the T.T. and W.F. Chao Chair in Chemistry and a professor of materials science and nanoengineering.
The Air Force Office of Scientific Research (FA9550-22-1-0526), the Department of Energy National Energy Technology Laboratory (DE-FE0031794) funded the research.
- Peer-reviewed research
-
Flash recycling of graphite anodes: https://onlinelibrary.wiley.com/doi/10.1002/adma.202207303
- Images for download
-

https://news-network.rice.edu/news/files/2022/12/1212_FLASH-1-web.jpg
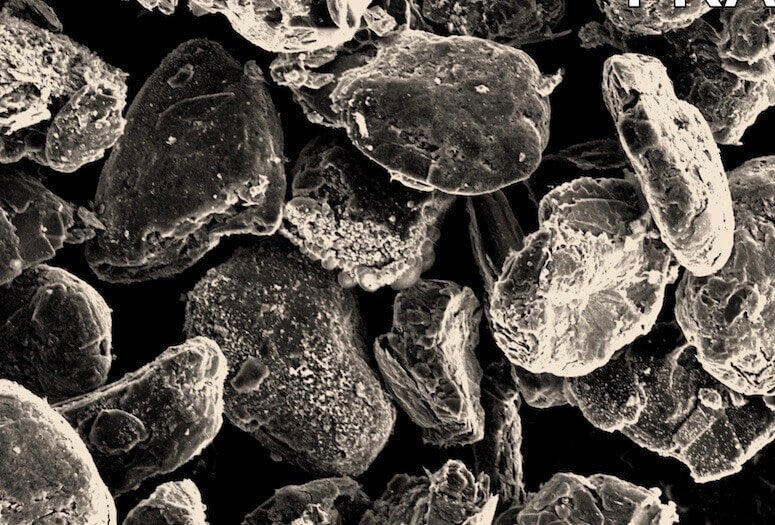
Flash-recycled anode particles as seen under through a scanning electron microscope. The particles are recovered from lithium-ion batteries and treated through Rice University’s flash Joule heating process. (Credit: Tour Group/Rice University)

https://news-network.rice.edu/news/files/2022/12/1212_FLASH-2-web.jpg
A transmission electron microscope image of a graphite anode particle recovered from a lithium-ion battery shows the carbon shell that forms around the graphite during flash Joule heating. The process developed at Rice University removes impurities from the spent anodes so the material can be reused. (Credit: Tour Group/Rice University)
https://news-network.rice.edu/news/files/2022/12/1212_FLASH-3-web.jpg
Rice University chemists use flash Joule heating to recover graphite anodes from spent lithium-ion batteries at a cost of about $118 per ton. (Credit: Weiyin Chen/Rice University)
- Related materials
-
Rice lab turns trash into valuable graphene in a flash: https://news.rice.edu/news/2020/rice-lab-turns-trash-valuable-graphene-flash
Flashing plastic ash completes recycling: https://news.rice.edu/news/2021/flashing-plastic-ash-completes-recycling
Machine learning fine-tunes flash graphene: https://news.rice.edu/news/2022/machine-learning-fine-tunes-flash-graphene
Cars could get a ‘flashy’ upgrade: https://news.rice.edu/news/2022/cars-could-get-flashy-upgrade
Tour Group: https://www.jmtour.com
Department of Chemistry: https://chemistry.rice.edu
Wiess School of Natural Sciences: https://naturalsciences.rice.edu
- About Rice
-
Located on a 300-acre forested campus in Houston, Rice University is consistently ranked among the nation’s top 20 universities by U.S. News & World Report. Rice has highly respected schools of Architecture, Business, Continuing Studies, Engineering, Humanities, Music, Natural Sciences and Social Sciences and is home to the Baker Institute for Public Policy. With 4,240 undergraduates and 3,972 graduate students, Rice’s undergraduate student-to-faculty ratio is just under 6-to-1. Its residential college system builds close-knit communities and lifelong friendships, just one reason why Rice is ranked No. 1 for lots of race/class interaction and No. 1 for quality of life by the Princeton Review. Rice is also rated as a best value among private universities by Kiplinger’s Personal Finance.