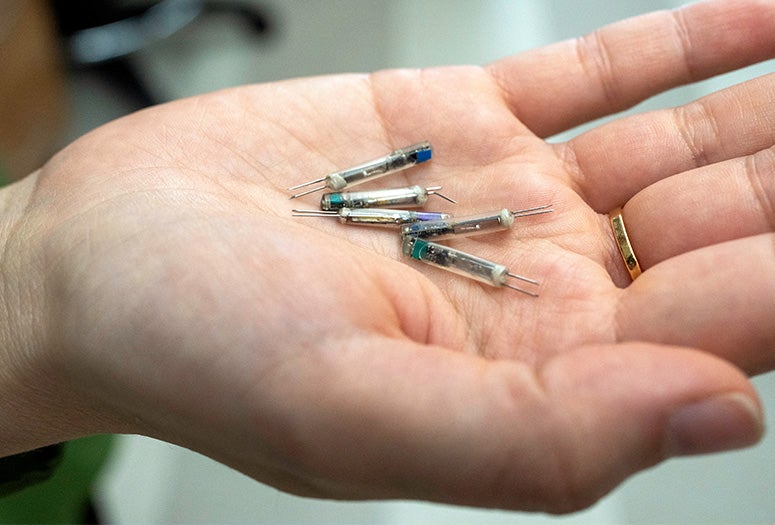
Researchers at Rice University and collaborators have developed a wireless network of miniature bioelectric implants that could transform treatment for heart failure, spinal cord injury and other chronic conditions. The system would integrate with patient anatomy easier than conventional medical implants, eliminating the need for batteries and invasive wiring.
In a new study published in Nature Biomedical Engineering, a team led by Rice engineer Jacob Robinson demonstrated that a network of tiny implants can deliver precise, distributed electrical stimulation to organs like the heart and the spinal cord while powered and controlled by a single external transmitter.
The system exploits the properties of magnetoelectric materials, which convert magnetic fields into electricity using mechanical vibrations. The magnetoelectric implants harvest energy from a magnetic field transmitter outside the body and deliver programmable stimulation pulses to surrounding tissue.
Notably, the greater the number of implanted units, the more efficient the system is. The research marks the first time such a system has been successfully deployed in large animal models ⎯ a critical milestone on the path to clinical use in humans.

“If we want to regulate patterns of electrical activity in nerves or in the heart or in the brain in a very precise and distributed manner, the challenge is reaching different depths and locations in the body ⎯ something that is very difficult to do with wired devices,” said Robinson, professor of electrical and computer engineering and bioengineering at Rice.
Traditional implantable medical devices like pacemakers and spinal stimulators are limited by a fundamental trade-off: More electrodes may allow for better, distributed stimulation, but this also entails more wires connecting them to a central power source and more invasive surgeries to implant and maintain.
The new Rice-developed system offers an alternative: a distributed network of devices precisely placed throughout the body, where each implant ⎯ roughly the size of a grain of rice ⎯ is individually programmable and responds to unique digital instructions from a single transmitter.
“Now we can go from one tethered device to many, making therapies more precise and adaptable,” said Joshua Woods, a Rice doctoral alumnus and a co-first author on the study.
Robinson, who is also the co-founder and CEO of Motif, a startup dedicated to commercializing a neuromodulation device that could revolutionize treatment for drug-resistant depression and other psychiatric or neurological disorders, said the research builds on his team’s previous work focused on miniaturizing devices and delivering power and data to them wirelessly.
“Up until now, we could not scale that technology beyond a single device,” Robinson said. “Developing a distributed network of devices meant figuring out how to make each node in the network individually addressable.”

This required adding more sophisticated electronics to the implants to enable them to respond separately to unique, individual messages embedded in the transmitter signal.
“The implants only respond when they receive digital sequences that are specific to each implant,” Robinson said. “It also turns out that system efficiency grows as we expand the size of the network. This feature of magnetoelectrics-based wireless power transfer is a key takeaway here.”
To test the implants in realistic clinical scenarios, the researchers used porcine models, which share many anatomical and physiological similarities with humans. In one experimental setup, four devices were implanted along the spinal cord, each targeting a different muscle group. When activated in sequence, the system successfully recruited distinct muscles in the hind limb ⎯ a technique with direct implications for paralysis rehabilitation and pain management.

In another setup, three wireless pacing nodes placed on the surface of a pig’s heart were used to consistently modify heartbeat. Coordinating activity across multiple points in the heart in order to resynchronize the heart’s rhythm, known as cardiac resynchronization therapy, is often limited by the number of leads physicians can thread into the heart. The performance of the wirelessly powered pacemakers in the Rice study suggests the platform could offer a less invasive and more customizable alternative with the potential for endovascular deployment through tiny catheters.
“A shared challenge for psychiatry, neurology and cardiac health is regulating patterns of electrical activity distributed over the entire organ, so the heart, brain and spinal cord are natural use cases for this technology,” Robinson said.
With further development, these networks could also include sensors, forming closed-loop systems that adjust stimulation in real time.
“This moves us closer to customizable bioelectronic therapies that can be placed exactly where they are needed in the body and personalized for each patient,” Woods said.
Robinson noted that strong collaborations with clinicians at the Texas Medical Center helped guide the research toward application areas with the greatest promise and facilitated clinically relevant testing.
“We were incredibly fortunate to partner with such outstanding clinical collaborators,” he said. “Their insight and support were critical in making these large-animal studies possible and in moving us closer to real-world use.”
Key collaborators included Philip Horner and Drs. Dimitry Sayenko and Amir Faraji at Houston Methodist and Dr. Mehdi Razavi at Baylor College of Medicine and the Texas Heart Institute (THI).

Razavi leads the Electrophysiology Clinical Research and Innovations lab at THI and is an associate professor of medicine-cardiology at Baylor College of Medicine, the program director for the Clinical Cardiac Electrophysiology Fellowship program at Baylor/THI and a member of the Rice Neuroengineering Initiative. He called the project “truly revolutionary.”
“This technology paves the way for remarkably innovative and highly impactful advances in the field of cardiac electrophysiology, the most important of which is enabling painless, imperceptible cardiac defibrillation,” Razavi said. “Defibrillator shocks, though lifesaving, can be traumatic for patients. By enabling the distribution of current across multiple pacing sites, Jacob’s work could vastly improve how patients experience these medical events.”
The research was supported by the National Institutes of Health (R01HL144683, 1R01NS119587-01A), the Walter Neurological Restoration Initiative sponsored by Paula and J.C. “Rusty” Walter III and the Walter Oil & Gas Corporation, the Wings for Life Foundation and the National Science Foundation. The content in this press release is solely the responsibility of the authors and does not necessarily represent the official views of funding organizations and institutions.
- Peer-reviewed paper:
-
Distributed battery-free bioelectronic implants with improved network power transfer efficiency via magnetoelectrics | Nature Biomedical Engineering | DOI: 10.1038/s41551-025-01489-3
Authors: Joshua Woods, Fatima Alrashdan, Ellie Chen, Wendy Tan, Mathews John, Lukas Jaworski, Drew Bernard, Allison Post, Angel Moctezuma-Ramirez, Abdelmotagaly Elgalad, Alexander Steele, Sean Barber, Philip Horner, Amir Faraji, Dimitry Sayenko, Mehdi Razavi and Jacob Robinson
https://doi.org/10.1038/s41551-025-01489-3 - Access associated media files:
-
https://rice.box.com/s/7p9z4qx26qmezqx6ys11yewilthofrhg
(Photos by Gustavo Raskosky/Rice University)