Rice scientists develop inorganic process for small-scale production
Rice University researchers have developed an inorganic method to synthesize ammonia that is both environmentally friendly and can produce the valuable chemical on demand under ambient conditions.
The Brown School of Engineering lab of materials scientist Jun Lou manipulated a two-dimensional crystal it understands well — molybdenum disulfide — and turned it into a catalyst by removing atoms of sulfur from the latticelike structure and replacing the exposed molybdenum with cobalt.
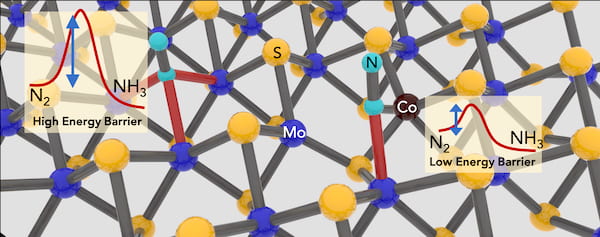
The addition of cobalt atoms to fill vacancies in 2D molybdenum disulfide crystals enhances the material’s ability to catalyze ammonia from dinitrogen. Rice University scientists have developed a “green” method for the small-scale synthesis of ammonia that uses less energy and produces less carbon dioxide than industrial processes. Courtesy of the Lou Group
This allowed the material to mimic the natural organic process bacteria use to turn atmospheric dinitrogen into ammonia in organisms, including in humans, who use ammonia to help liver function.
The inorganic process will allow ammonia to be produced anywhere it’s needed as a small-scale adjunct to industry, which produces millions of tons of the chemical each year through the inorganic Haber-Bosch process.
The research is described in the Journal of the American Chemical Society.
“The Haber-Bosch process produces a lot of carbon dioxide and consumes a lot of energy,” said co-lead author and Rice graduate student Xiaoyin Tian. “But our process uses electricity to trigger the catalyst. We can get that from solar or wind.”

Rice University graduate student Xiaoyin Tian, left, and postdoctoral researcher Jing Zhang led the effort to develop an inorganic catalyst for ammonia based on doped, two-dimensional molybdenum disulfide. Courtesy of the Lou Group
The researchers already knew that molybdenum disulfide had an affinity to bond with dinitrogen, a naturally occurring molecule of two strongly bonded nitrogen atoms that forms about 78% of Earth’s atmosphere.
Computational simulations by Mingjie Liu, a research associate at Brookhaven National Laboratory, showed replacing some exposed molybdenum atoms with cobalt would enhance the compound’s ability to facilitate dinitrogen’s reduction to ammonia.
Lab tests at Rice showed this was so. The researchers assembled samples of the nanoscale material by growing defective molybdenum disulfide crystals on carbon cloth and adding cobalt. (The crystals are technically 2D but appear as a plane of molybdenum atoms with layers of sulfur above and below.) With current applied, the compound yielded more than 10 grams of ammonia per hour using 1 kilogram of catalyst.
“The scale is not comparable to well-developed industrials processes, but it can be an alternative in specific cases,” said co-lead author Jing Zhang, a postdoctoral researcher at Rice. “It will allow the production of ammonia where there is no industrial plant, and even in space applications.” He said lab experiments used dedicated feeds of dinitrogen, but the platform can as easily pull it from the air.
Lou said other dopants may allow the material to catalyze other chemicals, a topic for future studies. “We thought there was an opportunity here to take something we’re very familiar with and try to do what nature has been doing for billions of years,” he said. “If we design a reactor the right way, the platform can carry out its function without interruption.”

Microscope images show cobalt-doped molybdenum disulfide as grown on a carbon cloth. The high-resolution transmission electron microscope image at right reveals the doped nanosheets, which facilitate the efficient electrochemical catalysis of ammonia. The process was developed for small-scale use by materials scientists at Rice University. Courtesy of the Lou Group
Co-authors of the paper are Rice assistant research professor Hua Guo and graduate student Qiyi Fang; Qin Wu of Brookhaven National Laboratory; and Jiadong Zhou and Zheng Liu of Nanyang Technological University, Singapore.
The Welch Foundation and the U.S. Department of Energy Office of Science supported the research.

