Rice University research suggests trigger for immune response and blood clotting
Rice University researchers have found an unexpected link between a protein that triggers the formation of blood clots and other proteins that are essential for the body’s immune system. The find could lead to new treatments for thousands of patients who suffer from inflammatory diseases and disorders that cause abnormal blood clotting.
The research is available online in the journal PLOS ONE.
“This link opens the door for studying severe, debilitating inflammatory disorders where the disease mechanism is still poorly understood, including lupus, rheumatoid arthritis, regional ileitis and ulcerative colitis, as well as age-related macular degeneration,” said study co-author Dr. Joel Moake, a hematologist and senior research scientist in bioengineering at Rice. “There’s clinical evidence that clotting and inflammation are somehow linked in many patients, even in the absence of an infection. This linkage could help explain some of the clinical cases that have long baffled physicians.”
The link is biochemical. Nancy Turner, a research technician in Moake’s lab, established the link after conducting hundreds of experiments on more than a dozen proteins, including key molecules involved with both clotting and the body’s innate immune response.

Rice University researchers Nancy Turner (left) and Joel Moake have found an unexpected link between a protein that triggers the formation of blood clots and other proteins that are essential for the body's immune system.
“In addition to the clinical evidence, there’s also a logical basis for this connection,” Moake said. “Clotting is a type of wound response, and wounds are magnets for infection, so there could be a selective advantage in triggering both responses at the same time.”
But the link could also have a downside. For example, if a person has a genetic mutation or acquired disorder that causes their blood to clot more often or more extensively than normal, the overactive clotting could lead to the kind of inflammation that would typically be caused by an infection. Furthermore, initiation of the clotting process may initiate clinical relapses in patients susceptible to various types of severe inflammatory disease.
In fact, the symptoms in the above scenarios are not uncommon. For example, in prior research, Moake’s lab conducted pioneering research on two disorders: thrombotic thrombocytopenic purpura (TTP), which causes clots to form in small blood vessels throughout the body; and hemolytic uremic syndrome (HUS), which causes abnormal blood clots in the kidneys. Both HUS and TTP come in two varieties — one that is triggered by infection or inappropriate antibody formation, and another that is hereditary. Moake said the newfound link between clot formation and the immune response could help improve the diagnosis and treatment for TTP, HUS and other puzzling blood disorders with similar symptoms.
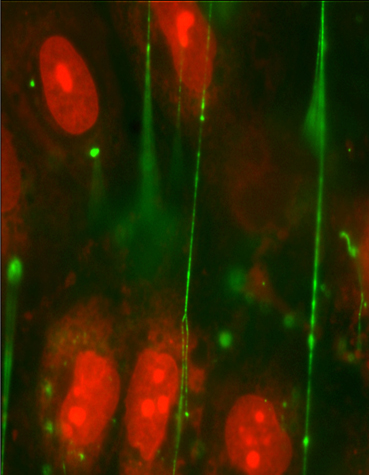
Von Willebrand factor (VWF) (green) is a long, thin protein that promotes blood clot formation. One end of VWF stays anchored to the blood vessel wall, while the rest unfurls like a sticky streamer, attracting platelets. CREDIT: Nancy Turner/Rice University
The experiments Turner used to establish the link between clotting and the body’s immune response involved a key clotting protein called von Willebrand factor (VWF) and about a dozen other proteins that are components of the “complement system.” The complement system, a part of the body’s innate immune system, is one of biology’s most ancient forms of defense against invading pathogens.
The complement system consists of a series of proteins that are produced by a variety of cell types. These proteins circulate continuously in the bloodstream and react sequentially upon activation. When triggered, the complement component proteins join together to form a biological weapon called the “membrane attack complex” (MAC), which kills both invading bacteria and the body’s own cells if they become infected or damaged.
Turner and Moake first thought of looking for the link more than two years ago after they collaborated with physicians at Texas Children’s Hospital on several puzzling clinical cases. Turner designed and conducted a series of experiments to examine whether any of the proteins in the complement complex were likely to bind onto long strands of VWF. Each complement protein was detected with a specific antibody and a fluorescent tag that could be viewed with a specialized microscope.
She found that C3, an important complement pathway initiator protein, was produced by cells in such low concentration that it was almost impossible to see — even with a fluorescent microscope. But that changed when she looked at experimental samples that contained both C3 and VWF.
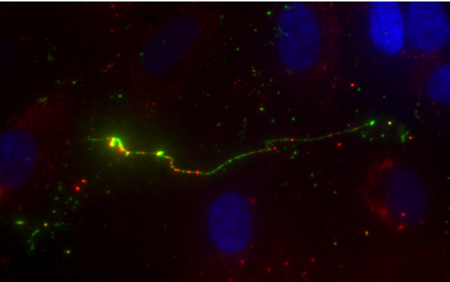
A strand of the clot-forming protein von Willebrand factor (VWF) glows brightly (green) under a microscope. The VWF is bound to many fluorescently labeled complement proteins called C3 (red dots), which are a part of the innate immune system. CREDIT: Nancy Turner/Rice University
“The signals were so clear,” Turner recalled. “The VWF had so much C3 on it that it looked like a Christmas tree.”
Moake said he and Turner are conducting follow-up research to measure more effectively the activation of C3 on VWF. They are also measuring whether the C3 activation stimulates the sequential cascade of reactions that leads to MAC formation. In particular, they are interested in studying how the connection might lead to autoimmune diseases by causing MAC to target the body’s own healthy cells rather than sick or damaged cells.
“We’d like to know what happens on a cell’s surface that ordinarily enables it to protect itself against MAC,” Moake said. “We’d also like to know what can go wrong with cells in terms of sickness or trauma that might make them more susceptible to being attacked and killed by overactivation of the complement sequence during clotting.”
The research was funded by the Mary Rodes Gibson Foundation and the Mabel and Everett Hinkson Fund.


Leave a Reply