Antibiotic study inspires new pathways in heart-valve research
By Shawn Hutchins
As people age, or as a result of poor nutrition, heart valves can become damaged by the accumulation of calcium deposits within the tissue. This calcification causes a thickening and hardening of the tissue to the point that it limits normal blood flow.

Pictured are bioengineering graduate student Dena Wiltz (left) and Hanszen College senior Aditya Kumar (back) with Associate Professor Jane Grande-Allen.
Bioengineering researchers in Rice Associate Professor Jane Grande-Allen’s laboratory analyze the biomechanics of heart-valve tissue and the underlying cellular and genetic causes of valve disease. One ongoing investigation pursued by her group looks into the roles cellular and matrix components play in normal valve biology and the degenerative processes that can cause the formation of calcified nodules in aortic valve leaflets.
“Very little is known about the intracellular dynamics among genes, proteins and the metabolic pathways that guide either normal processes or give rise to abnormalities,” Grande-Allen said. “Accumulative efforts in our lab involve investigations into valve-cell morphology and disease from a more mechanical perspective. Specifically, we look for cues as to how alterations to cells and cellular environments are driven by mechanical stress of the pumping heart over many years.”
In a unique twist of fate, Grande-Allen, graduate student Dena Wiltz and undergraduate student Aditya Kumar found that gentamicin, an aminoglycosidic antibiotic used to treat many types of bacterial infections, significantly reduced the number and size of calcific nodules formed by valvular interstitial cells.
The research, which was detailed in the journal Cardiovascular Engineering and Technology, was initially spurred by the lab’s examination of various antibiotics for the occasional prevention of bacterial infections in cell cultures.
“Contamination of cell cultures is a consistent issue for researchers, said Wiltz, a sixth-year graduate student in the Grande-Allen lab at Rice’s BioScience Research Collaborative. Wiltz’s research in valve disease is supported by a grant from Baylor College of Medicine.
“Impurities not only adversely affect results, but vast amounts of time and money can be spent managing bacterial contaminates,” Wiltz said. “Gentamicin is widely used for the in vitro prevention of cell- and tissue-culture contamination, but it has been reported to affect calcium levels in various cell types. So we decided to add it to our cultures — not to prevent contamination so much as to see if it regulated calcium mineralization by valve cells.”
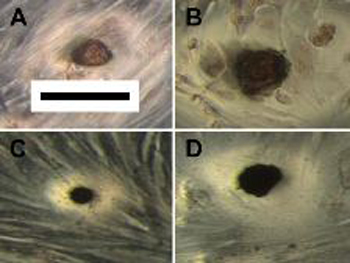
Varying images of calcific nodules stained in dark red with surrounding valvular interstitial cells in gray. Images B and D received the highest treatment of gentamicin.
Their investigations paid off. “Not only did we find that dose-dependent increases of gentamicin caused alterations to cellular mineralization, but the findings, along with a literary investigation, spurred insight into the roles cellular components, such as lipids and mitochondria, might play in the development of pathological calcification,” Kumar said. He is a senior and co-author on the research paper and has been a member of the Grande-Allen group since the summer of his freshman year.
Aortic valve tissue is made of trilayered connective tissues, and Grande-Allen and her students apply engineering analysis to decode how individual parts of this complex cooperate, respond to cellular signals and are influenced by overall valvular function, growth and the evolution of abnormalities.
“Through a bottom-up approach, we are looking at how valvular interstitial cells — the primary cells within these tissue layers — interact both biologically and mechanically with their surrounding environments,” Grande-Allen said. “Although gentamicin has limited clinical use, due to its toxic side effects, our results might prove to be useful in connecting these processes and ultimately in the development of promising pharmaceuticals that curb the progression of heart-valve disease.”
The research was also supported by a National Institutes of Health grant from the National Heart, Lung and Blood Institute.
– Shawn Hutchins is a Web coordinator and staff writer for the George R. Brown School of Engineering.


Leave a Reply